 And in aqueous solution,Īcetate anions react with water to form acetic acid and hydroxide ions. We need to think about the acetate anion in aqueous solution. So if our goal is to figure out the pH of the resulting solution, we don't need to consider acetic acid and we don't need toĬonsider the hydroxide ions 'cause these have been completely used up. Neutralize each other and form the acetate anion. Moles of our weak acid and our strong base and the mole ratio is one-to-one, the weak and the strong base are going to completely Net ionic equation, the mole ratio of our weakĪcid to our strong base is one-to-one. And the first situation, we have equal moles of our 
Now that we have our net ionic equation, we're gonna think about So for our net equation, we have acetic acid plus hydroxide anion, forms the acetate anion and water. After we take out the sodium cation, what's left over is So looking at the complete ionic equation, there are sodium cations on the left and sodium cations on the right, so we can cross out the sodium cation, that's our spectator ion. And to do that, we first need to identify spectator ions. Next, we're gonna use theĬomplete ionic equation to write the net ionic equation for this weak acid-strong base reaction. So this balanced equation represents the complete ionic equation. In some aqueous subscripts and a liquid one for water, some plus signs, and the reaction arrow. To an extremely small extent, we don't write it as the ions. Therefore, in aqueous solution, we would have sodium cations, Na+, and the acetate anion, CH3COO. So an aqueous solution of sodium hydroxide consists of sodium cationsĪnd hydroxide anions. Sodium hydroxide is a strong base, and strong bases dissociateġ00% in solution. However, things are differentįor sodium hydroxide. We're not gonna show acetic acid ionizing into H+ and the conjugate base. 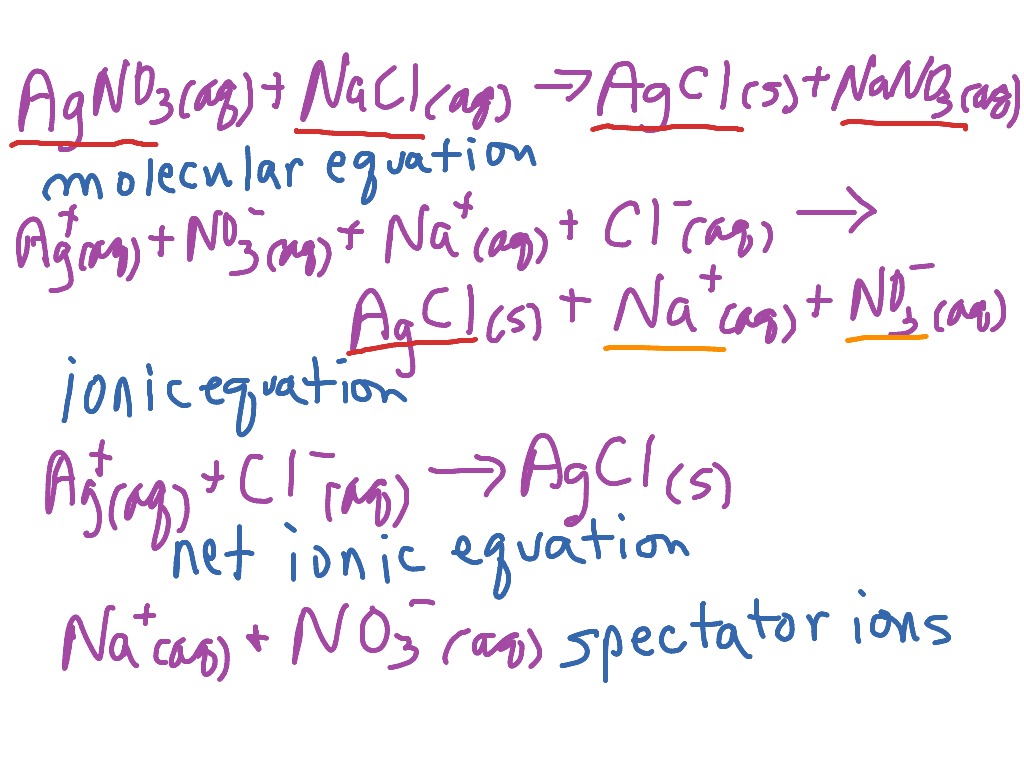
Molecules stay protonated and don't turn into acetate. 
Solution of acetic acid, most of the acetic acid Acetic acid is a weak acid that only partly ionizes in solution. This is also called theĬomplete ionic equation. Since this reaction is anĪcid-base neutralization reaction, and these reactions go to completion, instead of using an equilibrium arrow, we simply draw an arrow Sodium acetate is formed along with water. With sodium hydroxide, an aqueous solution of Is an example of a weak acid and sodium hydroxide is anĮxample of a strong base. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
